Approximately 130,000 knee replacement surgeries are performed every year in the United States. Too many of these knee replacements in recent years have had defective hardware.
Our Exactech Optetrak knee replacement lawyers are investigating the lawsuits and other potential defective knee replacement lawsuits throughout the United States for victims who received a defective knee implant. Call a knee replacement lawyer for a free consultation today at 800-553-8082 or get a free online consultation.
Knee Replacement Lawsuit Background
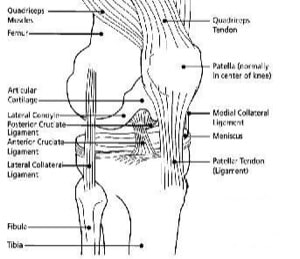
The knee is the largest joint in the human body. The knee joint supports the majority of the body’s weight, and as a consequence is susceptible to injury or trauma with the development of osteoarthritis in later life.
The human knee wears with age. Two of the primary causes of knee degeneration or wear are osteoarthritis and rheumatoid arthritis. With either of these conditions, the articular surfaces, which are normally covered with cartilage, wear out or degenerate. When these surfaces wear out, they expose the underlying bone surface and result in bone surfaces on the femur and tibia rubbing against each other. Because cartilage contains no nerve endings, healthy patients do not experience any sensation caused by an articulation of the femur and tibia.
When the cartilage degenerates, the bone-on-bone contact which occurs causes pain to the patient because the exposed bone surfaces contain nerve endings. Because physicians cannot replace cartilage, they use prosthetic devices to resurface the articular surfaces of the knee. Knee replacement surgery is often necessary for patients with osteoarthritis to alleviate pain when non-surgical therapies are ineffective.
An estimated 581,000 knee replacements are performed annually in the US. Direct costs of hip and knee replacements are greater than $35 billion. Over 4.5 million people have knee replacements. Is there a battle over that money by companies fighting to get to that market as quickly as they can? You bet. By 2030, it is projected that total joint replacement will increase to 4 million patients annually.
Various knee replacement devices are available and used in practice including polyethylene, metal, and ceramic knee replacement devices. First-generation knee implants were polyethylene, however, over time, these devices proved to be sub-optimal and as a result, manufacturers turned to metal-backed designs cemented to the tibia, and more recently cementless designs.
Manufacturers continue to research and bring to market devices that have lower rates of dislocation and increased longevity of the implant. Factors that impact long-term outcomes include the type of device implanted, the surgical technique, and patient factors including activity, age, gender, body mass, etc.
How Do Knee Implants Work?
Knee replacement surgery is a frequent choice for patients who are at their wits end from arthritis or extensive fractures. These patients did not want surgery but they just ran out of options. The following are the most common indications for surgery:
- Arthritis: inflammation of the knee joint which causes wear and tear on the knee cartilage.
- Osteoarthritis: the most common type of knee arthritis, and results in bone-on-bone contact at the knee joint.
In a total knee replacement surgery, surgeons remove the bone and cartilage on the end of the femur (thigh bone) and the shin bone (tibia). Devices, historically made of plastic but now more frequently, metal, are then attached to the bones to give the patient a new knee joint.
What Was Wrong With Smith & Nephew Implants?
First, let’s be clear. Our knee replacement lawyers are no longer taking Smith & Nephew claims. But Smith & Nephew had the first knee replacements that were a concern. They promoted their Journey knee devices as a device that potentially lasts longer than similar devices due to a lower rate of wear. Their knee replacement implants use oxidized zirconium rather than cobalt chrome or titanium which is more commonly used in replacement devices. Studies have shown that as cobalt devices wear, particles are released which can result in inflammatory reactions. Our knee replacement lawyers saw back then an industry that greased the wheels of future knee replacement lawsuits with a focus on profits and competition and the expense of patient safety.
Plaintiffs’ attorneys at first zeroed in their attention – and lawsuits – on Zimmer knee replacements This led both lawyers and researchers to take a closer look at knee replacements generally. What was found is that these new knee replacements are inferior, including the Smith & Nephew knee replacement (which led to the Optetrak Exactech and others). The common thread in these knee implant lawsuits was not the type of defect but the lack of care to make a safe knee replacement.
How did these knee replacements get on the market? Good question. Many joint replacement devices receive regulation under the 510(k) notification program. This FDA program allows medical devices to enter the market without requiring manufacturers to conduct clinical studies. Instead, the manufacturer must only demonstrate the device is substantially equivalent to other similar products already on the market. So the actual product that goes into a patient’s knee is not tested like these medical devices should be tested.
The FDA announced a Smith & Nephew recall of nearly 40,000 defective tibial baseplates in January 2010. An Alabama man filed a lawsuit against the manufacturer after receiving a knee replacement in 2008 which included a Journey tibial baseplate. The baseplate broke within 2 years of the replacement surgery. Now many other suits have followed against Smith & Nephew including one alleging that a lateral proximal tibia locking plate was defective and broke within 6 months of implantation and another case where Journey knee components completely failed within a year and a half, resulting in revision surgery.
As the population ages, this is only going to get worse. There will be more and more joint replacement surgeries with orthopedic implants leading the way. We just have to hold the makers of knee replacements accountable. A starting point? Conducting properly designed clinical trials to determine the safety and well-being of patients receiving these implants. This is a critical step that needs to be taken to reduce the necessity for revision surgery and in turn, alleviate the associated burden on the healthcare system. Will it happen? I think the question hinges on how successful these knee replacement lawsuits will be. Are we going to make these device makers make sure they get it right?
The Future
Exactech Optetrak, DePuy Attune, and Arthrex IBalance are knee replacements that are already causing problems. The Depuy Synthes’ Attune Knee System has received more than 230 FDA reports of early failure. In 2013, the Exactech went to a fit tibial tray on its Optetrak knee implant. This has led to a large number of revisions although that was just the start of the trouble for Exactech. DePuy, no stranger to high profile recalls, has had a difficult time with its Synthes’ Attune implant. Lawsuits are starting to be filed regarding these possible defective knee replacements.
The biggest target is DePuy Synthes, which DePuy Synthes, part of the Johnson & Johnson Medical Devices Companies, provides one of the most comprehensive orthopedics portfolios in the world. DePuy Synthes solutions, in specialties including joint reconstruction, trauma, craniomaxillofacial, spinal surgery, and sports medicine, are designed to advance patient care while delivering clinical and economic value to healthcare systems worldwide.
If you have a knee replacement lawsuit, there is a good chance you found out about it from a letter from your doctor telling you that you were given a defective knee implant. This is unlikely to be your doctor’s fault. Your doctor can only rely upon the information from the knee replacement manufacturer.
Example Exactech Knee Recall Lawsuit
Let’s take a look at a recently filed Exactech lawsuit. The civil case of Melissa Davis. v. Exactech Inc. (3:22-cv-1236), filed in the U.S. District Court for the District of South Carolina, is a very recent and typical example of an Exactech recall lawsuit involving premature failure of an Optetrak knee implant system. The plaintiff, Melissa Davis, is a resident of Leesville, South Carolina.
At 28 pages, the Complaint in the Davis case is a shorter version of many of the other Exactech recall lawsuit that have recently been filed. The Davis complaint recites word-for-word many of the same general factual allegations regarding Exactech’s development and marketing of the Optetrak implant system.
The Davis Exactech lawsuit alleges in very stark terms that Exactech continued to manufacture and sell the Optetrak knee implants for years despite being aware that the implants had an abnormally high rate of early failure:
Despite Defendants’ claims in its promotional materials of over 30 years of successful outcomes with knee devices, Defendants knew of an unacceptably high early failure rate of their Optetrak knee implants. On information and belief, Defendants concealed this information while its CEO and President David Petty bragged about the strong double-digit growth in knee business. From 2012 to 2016, a time when Defendants should have been recalling products, Exactech’s Extremities division almost doubled in size, from $52.1 million (2012) to $100.3 million (2016).
Davis Cmpl. ¶ 29.
Plaintiff, Melissa Davis, had her Opterak knee implant system installed as part of a total knee replacement surgery she underwent in 2017 at the age of 52. Her defective knee replacement lawsuit alleges that her Exactech knee implant failed prematurely and asserts a number causes of action including both traditional product liability claims (Negligence and Strict Liability), as well as consumer fraud and warranty claims.
Do I Have a Potential Knee Replacement Lawsuit?
We are investigating DePuy Attune, Exactech Optetrak and Arthrex IBalance and other knee replacements. Our primary focus in 2024 is on the Exactech Optetrak.
If you or a loved one has used these knee implants and experienced looseness, pain or discomfort from the implant, please call one of our knee replacement attorneys at 800-553-8082 or get a free no-obligation consultation on your potential knee lawsuit. Our knee replacement attorneys get no fee unless we get a recovery for you.
 Products Liability
Products Liability





